A cure for Alzheimer’s has arrived in 2022 as the newly discovered drug hailed a historic moment
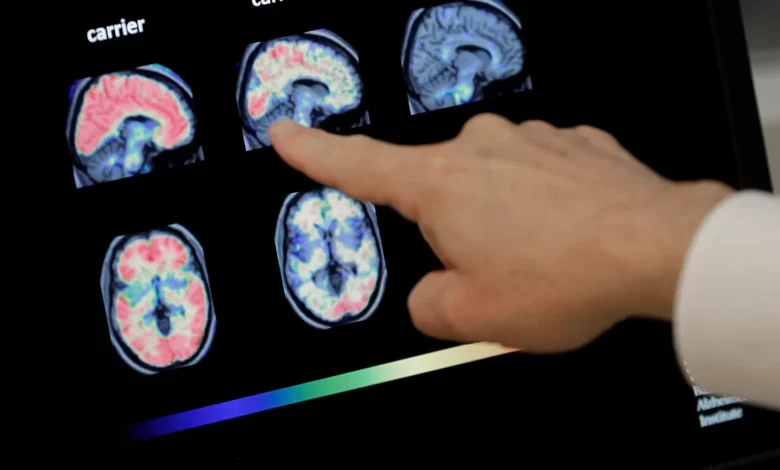
Japanese Drugmaker Eisai stated that cognitive decline in Alzheimer’s patients slowed in patients within 18 months who were administered the drug called lecanemab.
The Japanese drugmaker Eisai has revealed that the experimental drug for the treatment of Alzheimer’s disease helped to slow down the cognitive symptoms during the initial months of infection.
The company further revealed when a phase three trial was conducted, the drug was able to slow down cognitive decline by 27 percent after being administered for eighteen months. The results were released in a news interview and have not yet been subjected to discussion by the doctors.
The results may come as a bright ray of sunshine to Alzheimer’s patients after the U.S drugmaker Biogen’s ceased the supply of the drug. In addition, Biogen partnered with Eisai in the commercialization of the new drug.
However, Eisai has been credited for the development of the drug and for conducting the phase 3 trial.
Experts have requested that caution must be followed in interpreting the results of the drug. The results are the first step in the determination of the drug to make a significant impact on the disease.
The discussion was conducted by Dr. Ronald Peterson, a neurologist at the Mayo Clinic in Rochester, Minnesota.
A neurologist at the College of the University of Cincinnati has talked about the benefit of the drug, citing that the curing capacity was not so significant, and it might be below the threshold of what would prove to be beneficial for the patient.
The trials were conducted on 1795 patients who were randomly assigned to receive the drugs or their placebo for eighteen months.

The rate of cognitive decline was measured with the help of a clinical dementia rating scale that focused on six areas: memory, orientation, judgment and problem solving, community affairs, home and hobbies, and personal care.
The side effects of lecanemab and aduhelm were compared and found to be similar, resulting in brain bleeding and small swellings.
Brain swelling was reported in 12.5 percent of the patients who received the medication compared to 1.7 percent in the placebo group. The data was revealed by Eisai.
However, many patients displayed brain swelling and did not experience any symptoms. The prevalence of the side effect was lower than what was observed with other similar experimental drugs, as revealed by the drugmaker company.
Eisai has further stated that they would display the findings of the trial at an Alzheimer’s conference in late November and that it plans to submit the trial data to the Food and Drug Administration for approval by March.
They have even revealed that the FDA has already agreed to consider the drug under the organization’s accelerated approval pathway- the same process utilized to approve Biogen’s Aduhelm.
However, the decision for the conditional approval would be made by the agency in Early January.
The success of the drug could be a historic moment for the world.
Why is there no effective cure for Alzheimer’s?
Alzheimer’s is a degenerative brain disease. There are no effective cures or treatments to stop or slow down its development.
In addition, the disease has a drastic impact on 50 million in the world, thus resulting in dementia and death. Most individuals suffer from dementia which is not a normal part of ageing.
Constant research is conducted to overcome the disease. Recently, researchers of the University of Malaga, in collaboration with the University of Texas, have identified a possible treatment for Alzheimer’s disease.
It is believed that Alzheimer’s is caused by the increase in the toxic protein in the brain and ultimately results in neuronal death. In a preclinical model, the researchers have emphasized reducing the toxin protein.
They have stated that the toxic proteins are in equilibrium in the brain and the blood. However, the preclinical model is only used in paper and has not been proved yet.
The causes of Alzheimer’s have not been determined accurately despite decades of continuous research.
Consequently, it has become difficult to develop a drug with optimum effectiveness for the cure of illness. The first drug that met the requirements was the drug Aduhelm.
The drug was the first to address the cognitive decline in the disease.
However, the approval of the drug was a controversial statement. The clinical trial statements associated with the drug were ambiguous, with one trial showing positive results and the others not showing a similar trend.
In addition, the pricing of the drug was taken into account, claiming the drug to be excessively expensive.
Both Lecanemab and Aduhelm are designed to reduce the beta-amyloid plaques in the brain, and it is believed that the plaques are responsible for causing the disease.
The press release of the lecanemab quotes states that the drug is effective and proves the amyloid plaque hypothesis.
Eisai’s stock price further hiked by more than 60 percent after announcing the success of the drug in the trial.
It is now a matter of time to check if the drug would be a historic breakthrough in overcoming Alzheimer’s disease and if it can overcome the shortfall of the Aduhelm.
Edited by Prakriti Arora




