China approves genuine biotech’s HIV drug Azvudine for curing covid-19 patients

On Monday, China conditionally the domestic industry Genuine Biotech’s Azvudine pill, which is used to treat patients with Covid-19. The drug is proved to be another oral treatment option against the coronavirus. The treatment is currently limited to the usage of adults.
China’s top drug distributor has said that it has already granted the medicine to be used in the curing of the disease.

The availability of the covid-19 vaccines and treatments has proved to be crucial is important for the proper implementation of China’s ‘Zero Covid-19 policy’.The policy aims to eradicate every outbreak. However, the policy mainly emphasizes mass testing and strict quarantining protocols.
The Azvudine tables which have been approved by China in the previous year to cure certain HIV-1 virus infections have passed on the green signal to treat patients with the normal type covid. The reports were made by the National Medical Products Administration.
Normal type Covid is a term used by the Chinese people to describe a condition of coronavirus infections where there are signs of pneumonia. However, the patients have not reached a severe stage.
Previously in February, China recommended the use of Pfizer’s oral treatment Paxlovid for adults suffering from mild to high covid and with a high risk of developing a severe condition. In 2020, the Chinese government approved the use of the medicine Lianhuaqingwen capsules.
It is a traditional Chinese medicine formulation used for the treatment of influenza. The product is composed of more than a dozen of Chinese products that have been shown to significantly reduce the symptoms of Covid-19 by clearing excessive heat, removing toxins, improving lung ventilation, and discharging heat.
The manufacturers have stated that the home-grown covid-19 recovery drug has therapeutic potential and a safety profile for the treatment of patients infected with SaRS-CoV-2. The medicine mainly focuses on the efficacy and the safety of the medicine for Covid-19 infected patients. In addition, it emphasizes evaluating the clinical outcome of the Azvudine medicine in comparison to the patients infected with the severe stage of the virus.

In a late-stage clinical trial, 40.4% of the patients taking Azvudine medicine showed an improvement in their health condition after the consumption of the drug, compared with 10.9% in the group. However, Genuine Biotech has not provided detailed readings regarding the trial.
The company has even unveiled that the third and late-stage clinical trials show that oral medication is safe and can improve the clinical condition of the patients and eliminate the virus within five days.
The vice-president of Zhengzhou University and a leading researcher in the development of the pill has stated during an interview that the clinical trials conducted so far imply that the drug is effective against all the emerging variants of the covid-19 virus including Omicron.
Further, he added that the medication Azvudine’s price has not yet been determined. But, the price will be significantly lower than Pfizer’s Paxlovid pill, which costs 2,300 yuan (approx 340 USD) per dose.
Paxlovid was the first effective Covid-19 drug in mainland of China and the medicine was approved with conditional authorization in the country.
The first domestically developed Covid-19 medication available on the market was introduced by researchers from Tsinghua University and Brii Biosciences and later, approved in December. The drug is usually delivered via injection.
What are the composition, eligibility, and side effects of Azvudine- Genuine Biotechs formulation?
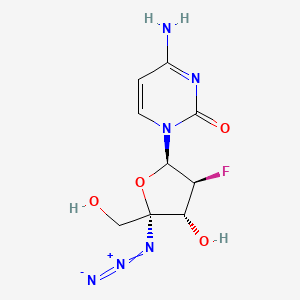
Azvudine (FNC) is a nucleoside analog that inhibits HIV-1 RNA-dependent RNA polymerase. Recently, it was seen that the azvudine analog was able to fight against the covid-19 virus when tested on a rat infected with the SARS-CoV-2 strain. The FNC monophosphate accumulated in the thymus and peripheral blood mononuclear cells.
A random single-arm clinical trial of the Azvudine drug showed that oral FNC cured all Covid-19 patients and 100% hospital discharge rates were recorded.
However, the administration of FNC is minor and followed by acute symptoms including nausea and dizziness. The symptoms were observed in 16.12% of patients.
Thus, scientists have concluded that the administration of the drug can prevent covid followed by increased immunity and the FNC is specifically concentrated in the thymus.
The formulation is recommended for the spectra of patients who fall under the following criteria:
- The patient’s age must be greater than 18 years and the administration of the medicine is irrespective of the gender.
- Symptomatic patients who meet the WHO case definition for Covid-19 with no signs of bacterial pneumonia or hypoxia.
- The medicine is restricted to patients with liver disorders or patients suffering from shortness of breath during the infection period.
- The medication can not be administered to pregnant as well as lactating mothers.
Can Azvudine drug compete with Pfizer’s drug Paxlovid despite costing less than the latter?
A comparison of Azvudine with Paxlovid revealed that the latter reduced the chances of hospitalization by a significant 89% in the patients who have taken the drugs for three days. Azvudine’s efficiency has been questioned by the mass and it is less effective than the Paxlovid drug. So, replacing the latter is not a possible attempt for the Genuine biotech firm anytime soon.
On the brighter side, Azuvdine’s drug would be available at cheaper prices and may prove to be a competition to paxlovid.
Edited by Prakriti Arora





