Four volunteers develop symptoms of Bell’s palsy post-Pfizer’s Covid -19 vaccine shot; Donald Trump okays rollout of First Shots in less than 24 hours
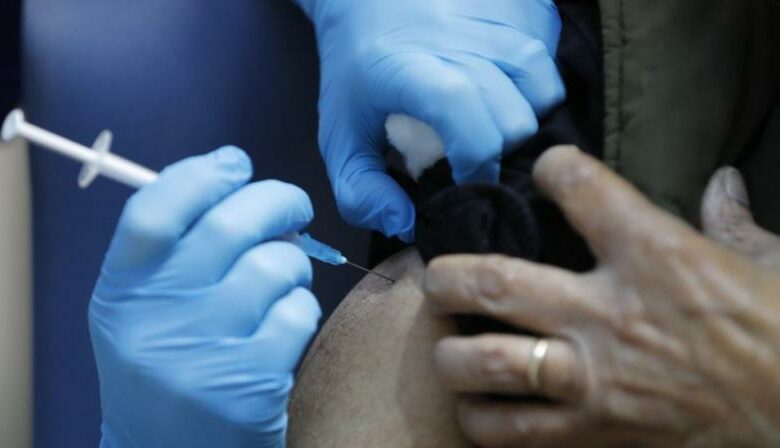
As Britain geared for inoculating its first set of volunteers for the coronavirus vaccine developed by Pfizer and BioNTech, the world waits in anticipation for both the vaccine’s success and the opportunity to get the vaccine themselves.
However, according to U.S. regulators’ report on the shots, four volunteers who were administered the first trial shots developed Bell’s palsy.
Bell’s palsy is a condition wherein it causes temporary weakness or paralysis of the facial muscles. It occurs when the nerves controlling the facial muscles become inflamed, swollen, or compressed.
This is a troubling side effect indeed, and FDA (Food and Drug Administration) regulators said that it is unclear as to what caused Bell’s palsy in the four volunteers and have asked the health experts to continuously monitor the condition of the volunteers who had participated in the first Covid -19 vaccine trials, and also watch out on how many people develop this side effect.
Ironically, Pfizer CEO Albert Bourla had earlier seemed unsure about the effectiveness of the vaccine. In one of his interviews with NBC’s Lestor Holt, he said, “I think this is something that needs to be examined. We are not certain about that right now with what we know”.
He also said that the Pharma giant had not “cut any corners” while rolling the vaccine out.
In his statement, he mentioned that the vaccine had been tested in the same way as testing any vaccine circulating out there. “This vaccine actually was tested, because of the scrutiny, with even higher standards in terms of how we do things.”
This may turn out to be a significant blow to the economically rich countries that are competing with each other to secure Covid -19 vaccines for their populations.
According to some reports, the US, Canada, and the U.K., among others, have secured enough Covid -19 vaccines for their entire population, and in multiple doses, this has obviously left the not so economic rich countries out of the vaccine race.
On the other hand, India cannot secure Pfizer and BioNtech Covid -19 vaccines since, as per India’s policies, any vaccine that can be given to the population needs to have trials for the same to have happened in the country. This means that for our people to get any vaccine, the tests for the same need to have been conducted in the country, and only then can the vaccines be made available for inoculation.
The news of Pfizer and BioNtech Covid – 19 vaccines was welcomed with a massive sigh of relief even as new coronavirus cases are being reported in many countries and coronavirus related deaths continue to climb worldwide with over 1.5 million casualties and more than 69 million cases.
However, on Tuesday, the documents released by U.S. regulators confirmed that despite few people developing Ball’s palsy’s side effect, the Pfizer vaccine was strongly protective against Covid -19 and appeared safe.
Meanwhile, Britain has started to inoculating the oldest citizens with shots of the Covid -19 vaccines.
Donald Trump says the rollout of the Pfizer and BioNTech Covid -19 vaccines in the next 24 hrs.
The (FDA) U.S. Food and Drug Administration approved the Pfizer and BioNTech Covid -19 vaccine for emergency use on Wednesday.
President Donald Trump has said that the vaccine rollout could begin in “less than 24 hrs and be free for all Americans.”
The United States remains to be one of the most affected by the Covid -19 pandemic, with nearly 16 million ( 1.6 crores) of its population affected by the coronavirus and about 300,000 (3 lakh) deaths owing to the Covid -19 pandemic.
The U.S. government also stated that they had struck a partnership with FedEx and UPS, which has enabled the government to begin shipping the vaccine to every state and zip code in the country, also adding that who will be administered the shots first will be at the discretion of the respective Governors of the state.
BioNtech readies more vaccine production for the United States.
The CEO of BioNTech, a German pharma company, has said that the biggest challenge that the company is facing along with its partner Pfizer is to scale the production of the vaccines as it has got a nod from the United States for the use of the Covid -19 vaccines which is a huge demand.
The company has in a statement said they would produce up to 1.3 billion doses of the vaccine next year, “We need to solve the manufacturing challenge, it is very clear that more doses are required, and we are dealing with the question – how to produce more doses,” Ugur Sahin said in an interview.
He also mentioned that the companies expect that conditional approval will be granted by the European Medicines Agency by the end of this month and can hence begin rolling out the vaccine in the European countries by next year.
The company has recently bought a plant from Novartis AG in Marburg, Germany, and hopes to meet the huge demand via starting manufacturing of the vaccines. It has said that it would begin making the vaccine there in the first half of 2021. Hence, they are working on getting the plant up and running on an expedited timeline.
The company plans to work on a baseline of 1.3 billion doses, and though not sure of how much it can expand the scale but have assured that it will try to do it significantly.
The company spokesperson also mentioned that the United States would receive and the supply will be limited at first in the U.S. The government has ordered 100 million doses of the two-dose vaccine and could negotiate for more doses.
However, in an interview with CNBC, the former FDA commissioner Scott Gottlieb also mentioned that the United States was offered to buy more doses as recently as last month by the company; however, United States had turned down the offer.
For now, as the race for Covid -19 vaccine gears momentum, it seems that the US, Britain, Canada, and the rest of the European countries are in the lead while the other countries of the world watch in awe and make plans as to how to procure the Covid -19 vaccines.




