India’s Serum Has Informed That 40 Million Dosages Of Astrazeneca’s COVID Immunization Are Ready And Are Planning To Take Up Novavax Shot Next

India’s Serum Institute has said it has made 40 million dosages of AstraZeneca‘s potential COVID-19 antibody. The ICMR will seek after “early accessibility” of the shot in India, it added. Serum Institute of India, the world’s biggest immunization maker, has said it has made 40 million dosages of AstraZeneca’s potential COVID-19 antibody, and will before long start making Novavax’s opponent shot, as the two of them look for administrative endorsement. Serum on Thursday said it had selected 1,600 members in India for late-stage preliminaries of AstraZeneca’s competitor and plans to look for administrative endorsement to run late-arrange preliminaries for the Novavax antibody. The AstraZeneca antibody, co-created by Oxford University, is the most developed in human testing in India, Serum stated, adding that the organization and the Indian Council of Medical Research (ICMR) will seek after “early accessibility” of the shot in India.
Delegates for both Serum and the ICMR declined to remark on whether the 40 million dosages of the AstraZeneca immunization were intended for flexibly just in India. Most recent COVID-19 antibody update. The ICMR, a central government body, had supported the clinical preliminary site expenses for the AstraZeneca immunization, Serum said. The organization and the ICMR are as of now directing mid-stage clinical preliminaries of the shot at 15 focuses across India. This news arrives at a time where coronavirus is overtaking India, the world’s second-most noticeably terrible hit nation, arrived at 8.68 million on Thursday and the loss of life expanded to more than 128,000. India likewise has at any rate two local Covid-19 immunization applicants being developed, while nearby drugmaker Dr. Reddy’s Labs is doing a preliminary in the nation for Russia’s antibody competitor.
‘Generally Advanced’:
The AstraZeneca-Oxford up-and-comer, considered one of the most encouraging Covid-19 antibodies, is being tried for viability in enormous preliminaries in the UK, Brazil, South Africa, and the US. Recently, Pfizer turned into the primary organization to deliver fruitful between time information for enormous scope Covid-19 immunization preliminaries, raising any desires for an early dispatch. In any case, no clinical preliminaries for the Pfizer antibody are being done in India, and nearby wellbeing specialists are placing their faith on a fast dispatch of the AstraZeneca-Oxford immunization. Prior to this week, Pfizer and its accomplice BioNTech said their antibody applicant had demonstrated over 90% compelling dependent on introductory preliminary outcomes. Pfizer said it was focused on propelling its commitment with India’s administration to gracefully its expected immunization in the nation.
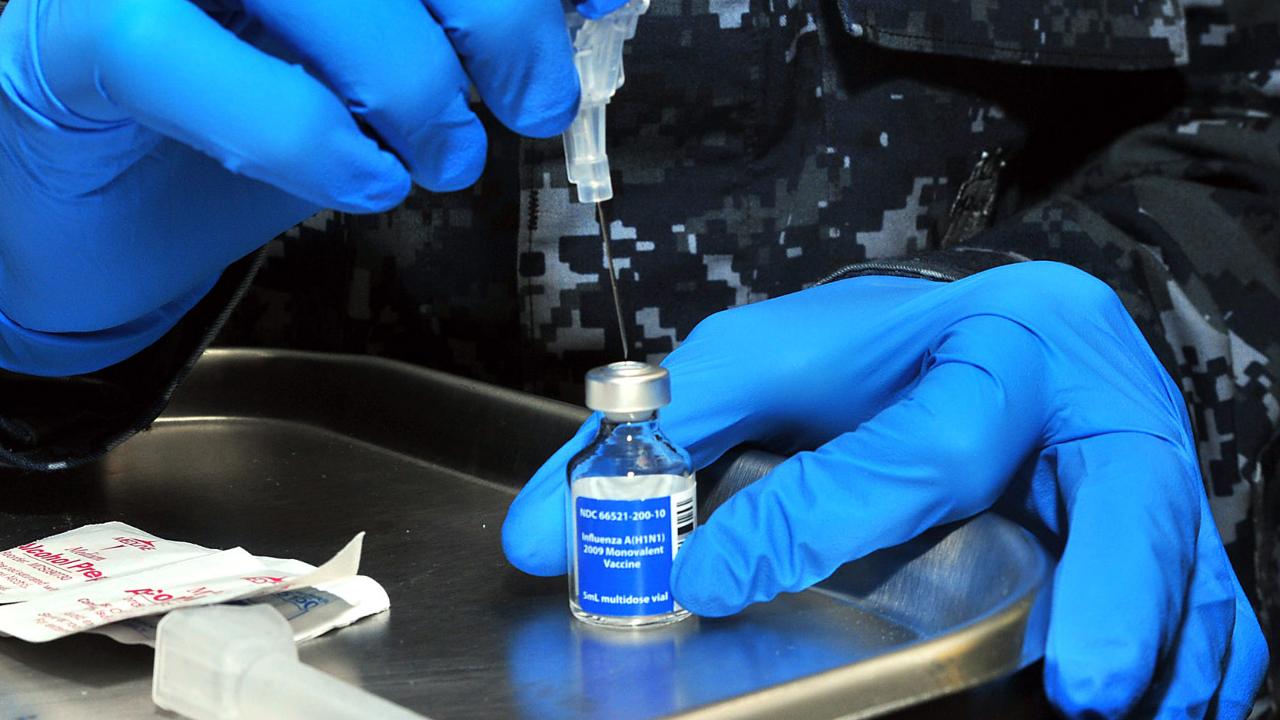 Serum Institute of India (SII) is inclining up the creation of AstraZeneca’s COVID-19 immunization, intending to have 100 million portions prepared by December for an inoculation drive that could start across India that very month, Bloomberg announced. If last stage preliminary information shows that AstraZeneca’s immunization applicant is giving compelling security from the infection, the Serum Institute of India – which is joined forces to deliver at any rate one billion portions – may get crisis authorization from New Delhi by December, said Adar Poonawalla, Chief Executive Officer, SII, in a meeting with Bloomberg. That underlying sum will go to India, Poonawalla said. Full endorsement right on time one year from now will permit appropriation on a 50-50 premise between the south Asian country and Covax, the World Health Organization-upheld body that is buying shots for helpless countries. An SII official disclosed to ET that if all works out positively, stage 3 preliminary outcomes will be accessible by December.
Serum Institute of India (SII) is inclining up the creation of AstraZeneca’s COVID-19 immunization, intending to have 100 million portions prepared by December for an inoculation drive that could start across India that very month, Bloomberg announced. If last stage preliminary information shows that AstraZeneca’s immunization applicant is giving compelling security from the infection, the Serum Institute of India – which is joined forces to deliver at any rate one billion portions – may get crisis authorization from New Delhi by December, said Adar Poonawalla, Chief Executive Officer, SII, in a meeting with Bloomberg. That underlying sum will go to India, Poonawalla said. Full endorsement right on time one year from now will permit appropriation on a 50-50 premise between the south Asian country and Covax, the World Health Organization-upheld body that is buying shots for helpless countries. An SII official disclosed to ET that if all works out positively, stage 3 preliminary outcomes will be accessible by December.
SII, which has tied up with five designers, has so far made 40 million dosages of AstraZeneca immunization in the previous two months and intends to begin producing Novavax’s competitor soon. Poonawalla is putting $250 million of his family’s fortune into an offer to increase producing ability to 1 billion portions through 2021. Then, SII additionally said it had gotten mass immunization and Matrix-M adjuvant from US-based Novavax, which it would fill into vials. The organization will before long apply for consent to lead stage 3 clinical preliminaries. Novavax has started late-stage clinical preliminaries in South Africa and the UK and is required to begin them in the US as well. One conspicuous immunization competitor requiring super chilly stockpiling is “a joke” that won’t work for the creating scene. Any individual who proclaims how long an antibody will give invulnerability is talking “hogwash.” The world’s whole populace won’t be inoculated until 2024, he stated, in opposition to rosier expectations.
 Poonawalla is similarly honest about the bet his organization is taking in the pandemic. The organization has redirected limit from existing immunizations and began chip away at another creation office to be finished one year from now at its central command in Pune. Poonawalla said the organization has sworn to keep half of the antibodies it makes for use inside India. It has just started fabricating the AstraZeneca immunization, he said. Around 20 million dosages have been made, and he hopes to have multiple times that sum prepared in the following four months, the report said. He is hopeful that in 2021, another Covid antibody will be authorized for public utilization each couple of months. For the creation of the Novavax NVAX.O antibody, Serum said it has gotten the main part of the immunization from the U.S. organization and will before long fill and finish them in vials. Novavax, which is trying its antibody in a late-stage concentrate in the UK, a month ago deferred the beginning of its U.S. preliminary by around a month because of deferrals in scaling up the assembling cycle.
Poonawalla is similarly honest about the bet his organization is taking in the pandemic. The organization has redirected limit from existing immunizations and began chip away at another creation office to be finished one year from now at its central command in Pune. Poonawalla said the organization has sworn to keep half of the antibodies it makes for use inside India. It has just started fabricating the AstraZeneca immunization, he said. Around 20 million dosages have been made, and he hopes to have multiple times that sum prepared in the following four months, the report said. He is hopeful that in 2021, another Covid antibody will be authorized for public utilization each couple of months. For the creation of the Novavax NVAX.O antibody, Serum said it has gotten the main part of the immunization from the U.S. organization and will before long fill and finish them in vials. Novavax, which is trying its antibody in a late-stage concentrate in the UK, a month ago deferred the beginning of its U.S. preliminary by around a month because of deferrals in scaling up the assembling cycle.

India likewise has at any rate two local COVID-19 immunization up-and-comers being developed, while neighborhood drugmaker Dr. Reddy’s Labs REDY.NS is doing a preliminary in the nation for Russia’s antibody applicant. Before this week, Pfizer PFE.N and its accomplice BioNTech 22UAy.DE said their immunization applicant had demonstrated over 90% compelling dependent on starting preliminary outcomes. Pfizer said it was focused on propelling its commitment with India’s administration to gracefully its likely antibody in the nation.




